The average number of new cases diagnosed each year was 2,965 in women and 20 in men. Breast cancer was the most common cancer in women in Ireland, accounting for 29% of all malignant neoplasms, excluding non-melanoma skin cancer (Table 4.1). During 1995-2007, the number of new cases diagnosed in women increased by approximately 3% per annum, 4% in RoI and 1% in NI. From 2002 to 2007, annual increases of 3% in RoI and 4% in NI were observed.
The risk of developing breast cancer up to the age of 74 was 1 in 12 for women and 1 in 1,621 for men and was slightly higher in RoI than in NI. At the end of 2008, 17,167 women and 53 men aged under 65, and 13,987 women and 128 men aged 65 and over, were alive up to 15 years after their cancer diagnosis.
Table 4.1 Summary information for breast cancer in Ireland, 1995-2007
Ireland | RoI | NI | ||||
females | males | females | males | females | males | |
% all new cancer cases | 21% | 0.1% | 21% | 0.1% | 21% | 0.1% |
% all new cancer cases excluding non-melanoma skin cancer | 29% | 0.2% | 29% | 0.2% | 28% | 0.2% |
average number of new cases per year 1995-2007 | 2965 | 20 | 1990 | 14 | 975 | 6 |
average number of new cases per year 1995-2001 | 2665 | 19 | 1762 | 12 | 903 | 7 |
average number of new cases per year 2002-2007 | 3315 | 20 | 2257 | 16 | 1058 | 4 |
cumulative risk to age 74 | 8.4% | 0.06% | 8.4% | 0.07% | 8.3% | 0.05% |
15-year prevalence (1994-2008) | 31154 | 181 | 20827 | 123 | 10327 | 58 |
The remainder of this chapter relates only to breast cancer in women.
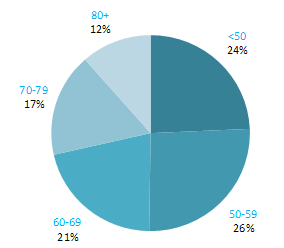
The proportion of breast cancers which occurred in women aged 60 and over was lower than for most other cancers (Figure 4.1). Almost one quarter of all cases occurred in women aged under 50, and a further quarter in those aged 50–59. Just 12% of cases were diagnosed in those aged over 80 years. This pattern was similar for RoI and NI.
Figure 4.1 Age distribution of female breast cancer cases in Ireland, 1995-2007
The incidence of breast cancer in women in RoI and NI was close to the median of the 21 countries shown. Age-standardised rates in both countries were slightly lower than in the UK (Figure 4.2). Rates were highest in Belgium and Denmark and lowest in Poland, Russia and Japan.
Figure 4.2 Estimated incidence rate per 100,000 in 2008 for selected developed countries compared to 2005-2007 incidence rate for RoI and NI: female breast cancer
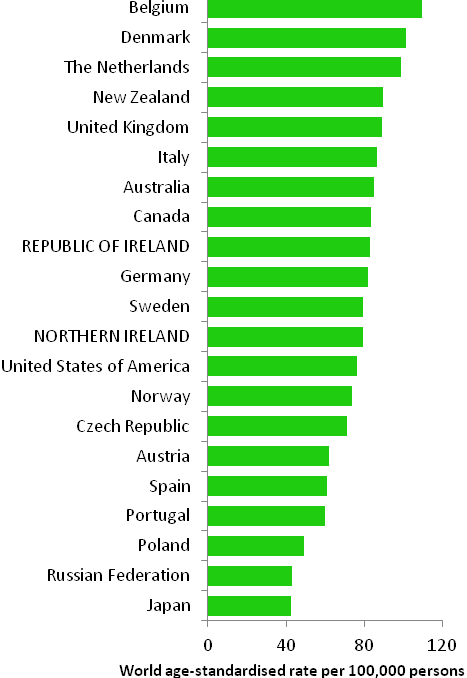
Source: GLOBOCAN 2008 (Ferlay et al., 2008) (excluding RoI and NI data, which is derived from cancer registry data for 2005-2007)
Table 4.2 Risk factors for breast cancer, by direction of association and strength of evidence
| Increases risk | Decreases risk |
Convincing or probable | Family history of breast cancer1,2 | Breastfeeding18,19 |
| Nulliparity and low parity2,3 | Physical activity18 |
| Late age at first pregnancy2,3 | Greater body fat (pre-menopausal cancer)18 |
| Late natural menopause2,3 | Tamoxifen and raloxifene5,20,21 |
| Early menarche2,3 | |
| Oral contraceptives4,5 | |
| Hormone replacement therapy5 | |
| Diethylstilbestrol5,6 | |
| Greater body fatness, abdominal fatness and weight gain in adulthood (post-menopausal cancer)7,8,9 | |
| Alcohol10,11 | |
| Smoking11 | |
| Ionizing radiation12, 13 | |
| Benign breast disease14 | |
| High socio-economic status15 | |
Possible | Red meat (pre-menopausal cancer)16,17 | Dairy food22 |
| Higher (own) birthweight 17 | Isoflavones from soya foods23 |
Vitamin D24,25,26 | ||
Dietary fibre27 | ||
Aspirin and other non-steroidal anti-inflammatory drugs28,29 | ||
1 First degree relative(s) with breast cancer; 2 Veronesi et al., 2005; 3 Key et al., 2001; 4 combined oestrogen-progestogen formulations; 5 International Agency for Research on Cancer, 2011a; 6 exposure during pregnancy; 7 World Cancer Research Fund / American Institute for Cancer Research, 2007; 8 Suzuki et al., 2009; 9 Vrieling et al., 2010; 10 Suzuki et al., 2008; 11 Secretan et al., 2009; 12 El Ghissassi et al., 2009; 13 Jansen-van der Weide et al., 2010; 14 Zhou et al., 2011; 15 Faggiano et al., 1997; 16 Taylor et al., 2009; 17 Xu et al., 2009; 18 International Agency for Research on Cancer, 2002; 19 Collaborative Group on Hormonal Factors in Breast Cancer, 2002; 20 in pre-menopausal women at high breast cancer risk; 21 Wickerham et al., 2009; 22 Dong et al., 2011a; 23 Dong & Qin, 2011; 24 intake and blood levels; 25 Chen et al., 2010; 26 Yin et al., 2010; 27 Dong et al., 2011b; 28 Takkouche et al., 2008; 29 Zhao et al., 2009 | ||
Breast cancer is a heterogeneous disease, comprising several distinct subgroups defined on the basis of hormonal receptor status and/or morphology. Recently interest has grown in distinguishing between risk factors for different subtypes (see, for example, Suzuki et al., 2008; Reeves et al., 2009; Suzuki et al., 2009; Vrieling et al., 2010; Yang et al., 2011). Up to 10% of breast cancer cases are hereditary and a woman's chance of developing the disease is increased if any of her first degree female relatives had breast cancer, particularly if more than one relative was affected at a young age (Veronesi et al., 2005). By age 70, women who carry BRCA1 gene mutations have a 65% chance of developing breast cancer, while those who carry BRCA2 mutations have a 45% risk (Antoniou et al., 2003). Family history may interact with other factors to modify risk, for example, exposure to low doses of radiation such as x-rays (Jansen-van der Weide et al., 2010) or history of benign breast disease (Zhou et al., 2011). Other than genetic factors, the major determinant of breast cancer risk is lifetime exposure to oestrogen (Table 4.2). Higher endogenous oestrogen exposure, as well as exogenous oestrogens, increases risk. In contrast, in pre-menopausal women at high risk of breast cancer, the anti-oestrogenic drugs tamoxifen and raloxifene reduce the chances of developing the disease by about half.
| Figure 4.3 Adjusted relative risks (with 95% confidence intervals) of breast cancer by socio-economic characteristics of geographic area of residence: females | |
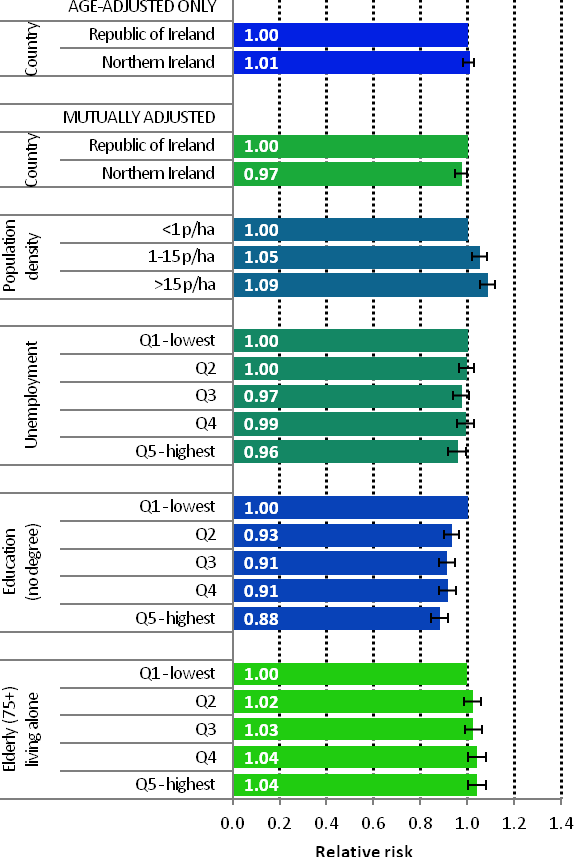 | The risks of breast cancer in women in RoI and NI were similar in 1995-2007; however, after adjustments for population density and socio-economic factors, the risk of breast cancer was lower in NI than in RoI (RR=0.97, 95%CI=0.95-1.00) (Figure 4.3) The risk of breast cancer increased with increasing population density. Women resident in areas with 1-15 persons per hectare (p/ha) had a 5% greater risk of breast cancer than those resident in the least densely populated areas, while those resident in the most densely populated areas had a 9% greater risk. Breast cancer risk was inversely related to both unemployment and educational attainment. Compared to areas with low levels of unemployment and high levels of degree level education, those areas with high levels of unemployment and less degree-level education, had a 4% and 12% lower risk of breast cancer respectively. Areas with the highest proportion of elderly living alone had a 4% greater risk of breast cancer. |
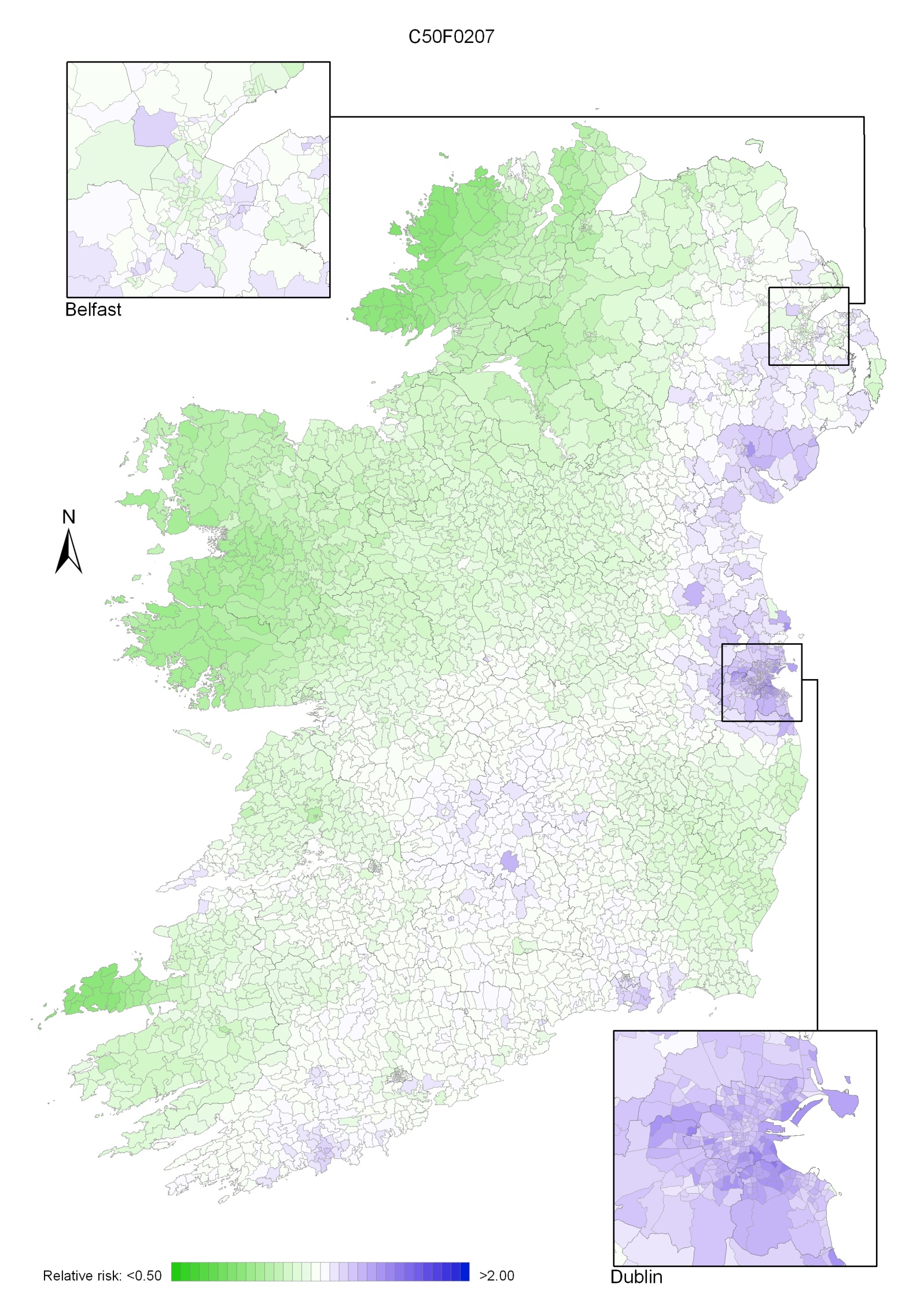
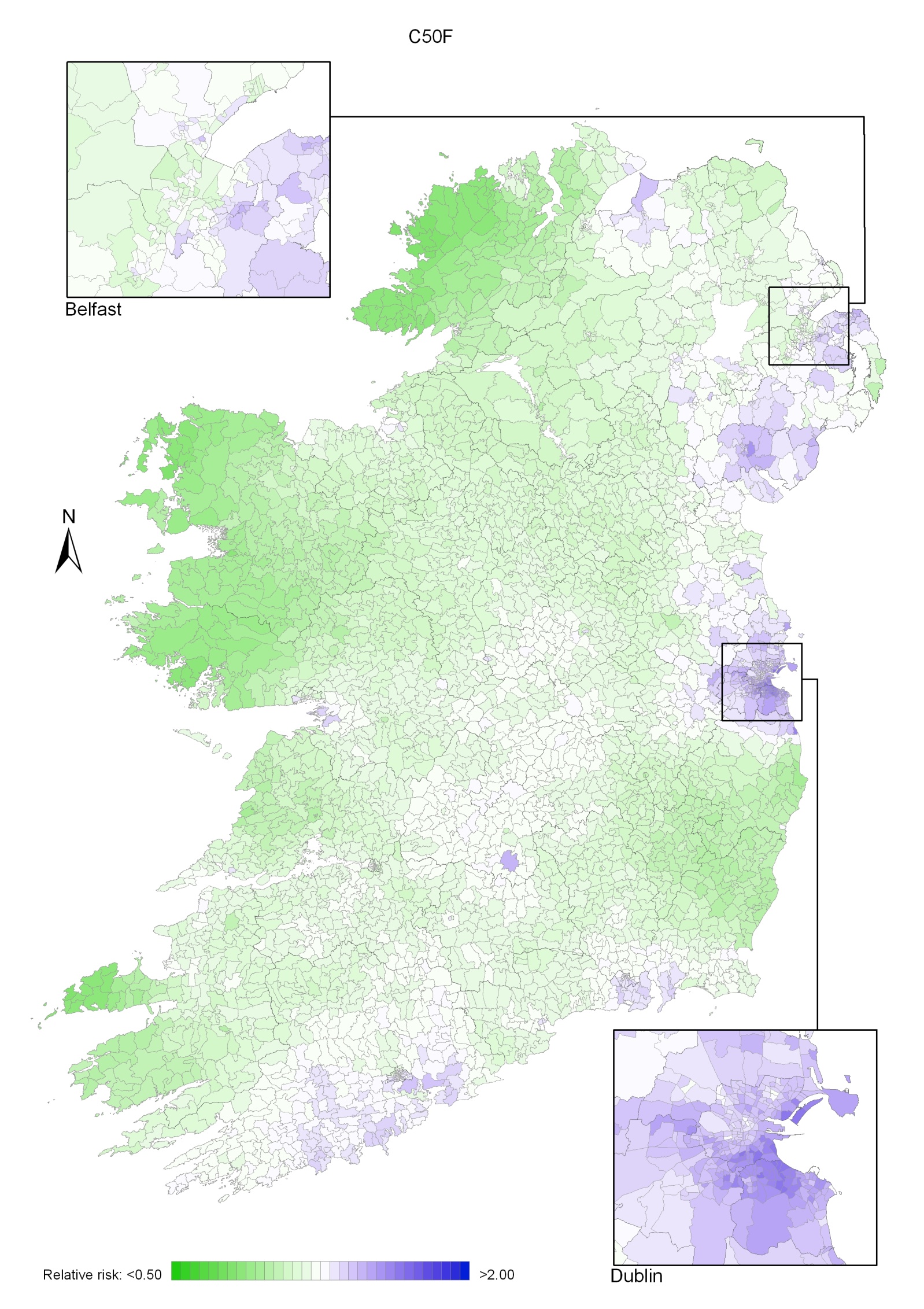
The geographical variation in relative risk of breast cancer was fairly modest (Maps 4.1-4.3).
Taking 1995-2007 overall, areas of higher relative risk existed around Dublin and Newry and Mourne and, to a lesser extent, Down, North Down, Ards, Limavady, and much of Munster (Map 4.1).
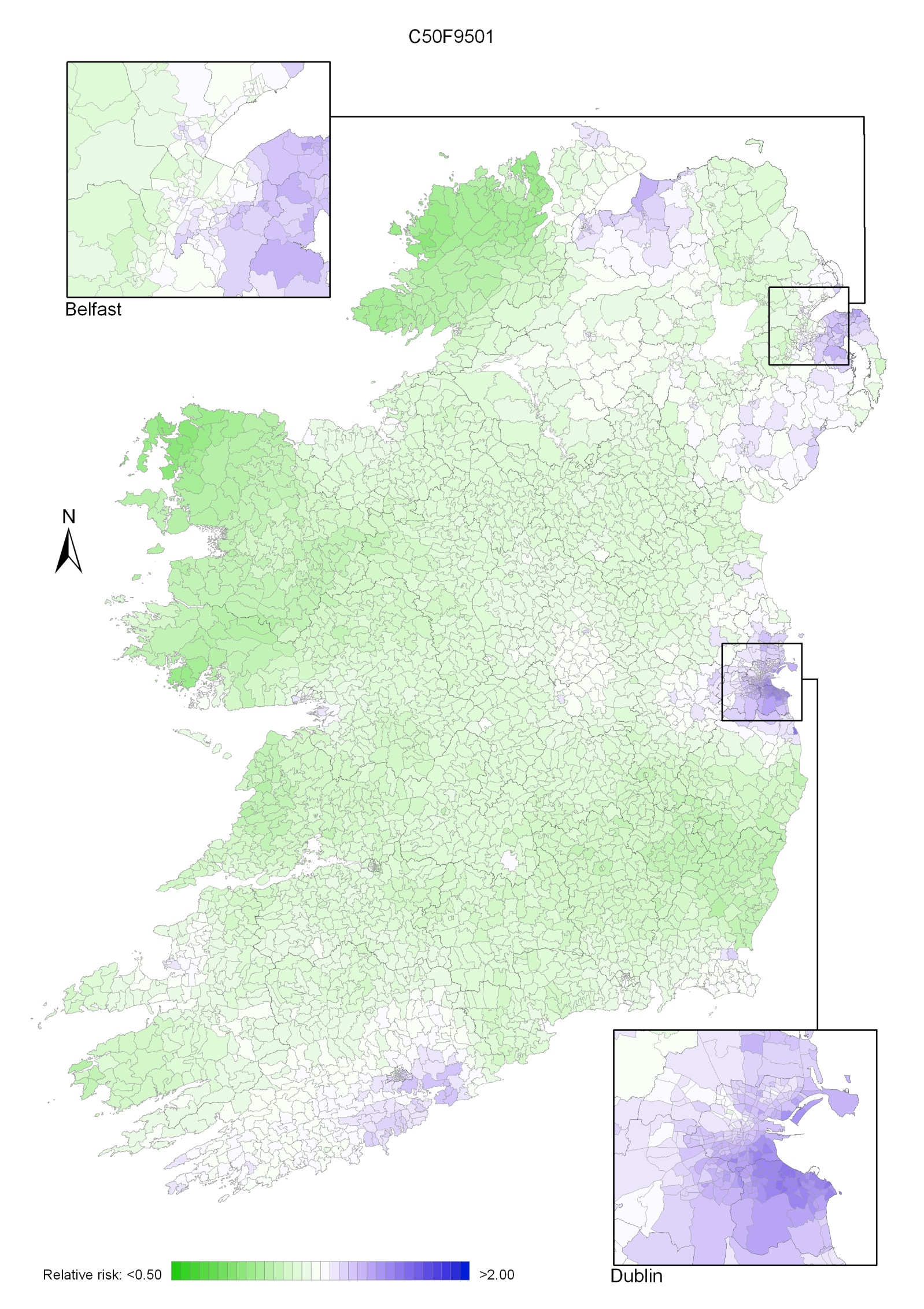
In the period 1995-2001, the areas of higher risk were in the major urban areas of east Belfast (including North Down), Dublin, Cork and Derry, and also in Limavady, Down, Ards and Castlereagh (Map 4.2).
In 2002-2007 the variation was somewhat more pronounced, with areas of higher relative risk extending in a band from Newry and Mourne to Dublin (Map 4.3). Areas in the west and north-west had consistently low relative risks throughout.
Map 4.1 Breast cancer, smoothed relative risks: females 1995-2007
Map 4.2 Breast cancer, smoothed relative risks: females 1995-2001
Map 4.3 Breast cancer, smoothed relative risks: females 2002-2007